
Assessing WBC Deformability
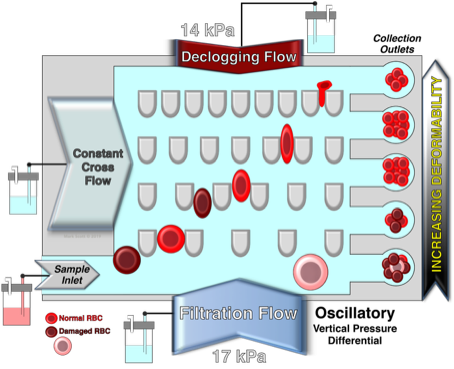
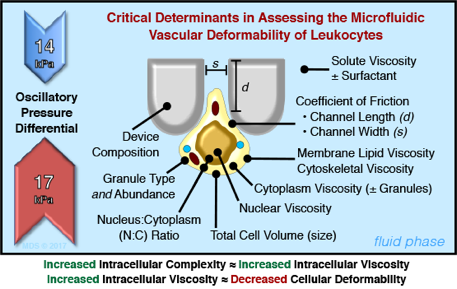
Ratchet Microfluidic Device
"...but be very careful with the WBC!"
"Please DO Squeeze the RBC..."
Despite the critical importance of mechanical (rheological + extrudability) deformability in the vascular flow of lymphocytes, it has been poorly investigated due to the limitations of existing technological tools. Microfluidics analysis of leukocyte deformability offers significant advantages in that it offers high throughput, large sample population and the ability to analyze a heterogeneous population. These advantages are in stark contrast to previous approaches that focused on single cell measurements. Importantly, the flow characteristics of microfluidic devices more closely model vascular deformability in that shear stress is applied forcing leukocyte passage through micropores of designed size. The modeling of vascular flow has been further enhanced by the development of a microfluidic ratchet device that introduced an oscillatory flow. As demonstrated in this study, the microfluidic ratchet device was able to separate human peripheral blood leukocyte subsets (i.e., monocytes and lymphocytes) based on differential deformability profiles. Furthermore, morphologically similar lymphocyte subsets (CD4, CD8 and NK) could also be separated. The subset separation was observed to be largely due to differences in their intracellular complexity (i.e., granule content) with granule-positive T lymphocytes and NK cells being less deformable than granule-negative lymphocytes. Moreover, upon immune activation, deformability of the de-granulated lymphocytes increased consequent to the decrease in cytoplasmic granularity/viscosity.
This study demonstrates for the first time that leukocytes subsets have differential deformability profiles and that intracellular granularity/degranulation significantly impacts the lymphocytes’ mechanical properties. These findings could be of clinical value as biomarkers of lymphocyte activation state and potential disease processes.
Mr. George Whipple: Charmin Toilet Paper spokesperson from 1964-1985, 1999-2000 and 2007. Appeared in over 500 unique commercials:
“Please, Don’t Squeeze The Charmin”
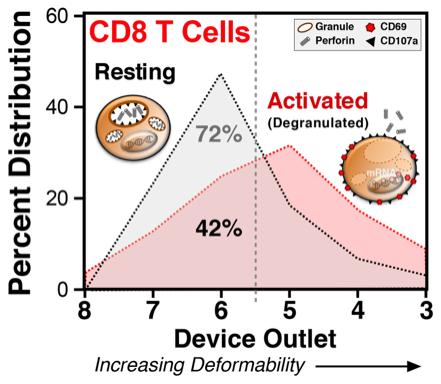
Activation-induced granule exocytosis led to increased CD8 T-cell deformability. Human PBMC with/without PMA + ionomycin activation were loaded and sorted on the microfluidic device. Cells from different outlets were separately collected, stained for T cell markers, and analyzed by flow cytometry. Activated CD8 T cells became more deformable and consequently showed a different microfluidic separation pattern from resting (unactivated) CD8 T cells.
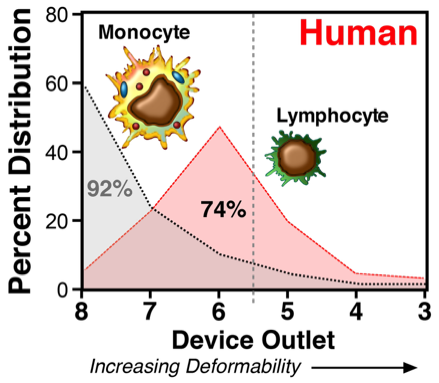
Monocytes and lymphocytes differentially sort in the microfluidic device. Monocytes, with larger cell sizes and more cellular complexity, primarily distributed in outlet 8 (pore size: 6.5 μm), whereas the relatively smaller and less complex lymphocytes were preferentially segregated among outlet 7-5 (blocking pore sizes: 5.5-4 μm) and peaked at outlet 6 (blocking pore size: 4.5 μm). The dashed line denoted between outlets 6 and 5 (4.5 and 4.0 µm funnels) represents the peak collection outlet for normal unactivated human lymphocytes. Values shown to the left of the dashed line are the mean percentage of total cells under the curve (100%) collected in outlets 8-6. Shown are the mean ± SEM from 3 different donors. Significance between monocytes and lymphocyte distributions in the individual outlets is shown (***p < 0.001; ****p < 0.0001).
Kang, N., Guo, Q., Islamzada, E., Ma, H., and Scott, M.D., Microfluidic Determination of Lymphocyte Vascular Deformability: Effects of Intracellular Complexity and Early Immune Activation. Integrative Biology, 10:207-217 (2018). DOI: https://doi.org/10.1039/c7ib00191f
Kang, N., Guo, Q., Islamzada, E., Ma, H., and Scott, M.D., Microfluidic Determination of Lymphocyte Vascular Deformability: Effects of Intracellular Complexity and Early Immune Activation. Integrative Biology, 10:207-217 (2018). DOI: https://doi.org/10.1039/c7ib00191f
Our microfluidic work is done in collaboration with Dr. Hongshen Ma at the University of British Columbia.
Dr. Ma is responsible for the design and manufacturing of the described microfluidic devices.
More information about Dr. Ma can be found at HERE.


